Features
 Prof. Claire Nichols, Associate Professor at the Department of Earth Science
Prof. Claire Nichols, Associate Professor at the Department of Earth ScienceJack is still actively studying the geology of the lunar surface, and when the Lunar Reconnaissance Orbiter which launched in 2009 took exceptionally high-resolution photographs of the lunar surface, he realised the true significance of some of the samples he had collected.
Rather than representing debris strewn across the Moon during meteorite impacts he realised that the boulders he had sampled, which are part of large-scale lava flows like those we see today in Hawaii, had barely moved since they cooled and crystallized over 3.5 billion years ago.
Prof. Claire Nichols, Associate Professor at the Department of Earth Science, studies planetary magnetic fields, and the Moon’s magnetic field history is particularly curious. Magnetic fields are driven in the dense iron-rich cores of planets by vigorous stirring of the electrically conductive, liquid metal.
Earth’s core is roughly half the radius of the planet, and the outer liquid part drives the bar-magnet-like field we observe around Earth today, responsible for the north and south magnetic poles aligned roughly along the planet’s rotation axis.
‘During the final manned mission to the Moon 49 years ago, Apollo 17 collected rocks from the lunar surface including samples which would turn out to be critical for our study nearly 50 years later.’
The Moon on the other hand has a tiny core, just one seventh of its radius. With such a tiny source of conductive material so far from the planet’s surface (the strength of the magnetic field drops off with distance from source cubed), we would never predict that the Moon generated a significant magnetic field. However, samples from the Apollo missions reveal that the Moon generated an intense magnetic field for at least 2 billion years.
Modellers have tried to simulate how such a strong and long-lived lunar magnetic field could be generated but have struggled to come up with plausible mechanisms. One issue is that observations have only revealed the strength of the magnetic field over time, rather than its geometry and orientation. Both observations are needed to be able to distinguish between the possible physical processes which stirred the molten part of the lunar core.
Almost all Apollo samples collected are loose material scattered across the lunar surface, and their original position and orientation are unknown. So when Jack realised he had collected samples whose original orientation could be recovered, he got in touch with the MIT Paleomagnetism Laboratory, to let us know he had just the samples we had been looking for.
After arriving on Earth, every sample from the Apollo missions was meticulously documented and photographed under lunar lighting conditions. In other words, light is shone onto the samples in such a way to exactly recreate the illumination and shadows when the sample was collected on the lunar surface.
‘The results suggest that the ancient lunar magnetic field had a very similar geometry to the magnetic field around the Earth today, which wasn’t what we had expected.’
We were able to borrow samples from the Johnson Space Center in Houston, Texas which were hammered off the rocky outcrops that Jack had determined must be close to their original position. We used the laboratory photographs to match up all the features with those in the astronaut photographs to work out the exact orientation the samples were in on the lunar surface when they crystallized 3750 million years ago.
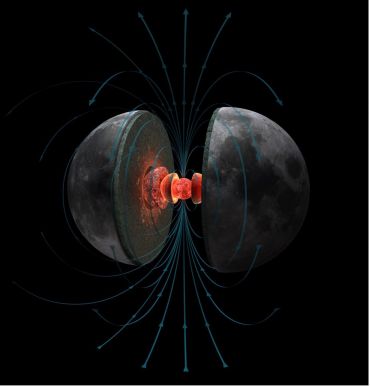 Magnetic fields are driven in the dense iron-rich cores of planets by vigorous stirring of the electrically conductive, liquid metal. Hernán Cañellas for the MIT Paleomagnetism Laboratory
Magnetic fields are driven in the dense iron-rich cores of planets by vigorous stirring of the electrically conductive, liquid metal. Hernán Cañellas for the MIT Paleomagnetism LaboratoryOnce we knew the orientation of the samples, we demagnetized them in the MIT Paleomagnetism Laboratory using a superconducting rock magnetometer to recover the strength – and for the first time – the direction of the ancient lunar magnetic field.
As previous studies had shown, we found evidence that the lunar magnetic field at this time was very intense – similar to the strength of Earth’s magnetic field today. We were also able to recover how steep the magnetic field was relative to the surface at Camelot Crater, where the samples were collected. This steepness can be used to determine where the magnetic north and south pole were relative to our sample’s location. We found that the magnetic poles were aligned along the present-day spin axis of the Moon (the axis around which the Moon rotates so that we always see the same side of it as it orbits around the Earth).
Our results suggest that the ancient lunar magnetic field had a very similar geometry to the magnetic field around the Earth today. This wasn’t what we had expected; given the strength of the Moon’s magnetic field, calculations show that it cannot have been generated by the same mechanism that drives Earth’s magnetic field. Additionally, because the Moon rotates incredibly slowly compared to the Earth - it rotates just once every 27 days - we wouldn’t expect the magnetic poles to be aligned along the spin axis. Instead, we would expect the magnetic field to be disorganized and have a much more complex pattern of magnetic field lines.
The alignment of the lunar magnetic poles along the spin axis is an important observation for understanding the interior dynamics of the Moon during its early history. Some model predictions have suggested that the mixing of molten iron in the centre of the Moon could have been physically driven by gravitational stirring. The rocky outer part of the Moon, the liquid metallic outer core and the solid metallic inner core all rotate at slightly different angles. This generates friction between the layers that could have generated a magnetic field – and given the alignment with the spin axis – seems the most likely candidate for driving the ancient field.
‘Samples from the Apollo missions reveal that the Moon generated an intense magnetic field for at least 2 billion years.’
The Moon is also thought to have shifted relative to its present-day spin axis over time. In other words, if we could travel billions of years back in time, we would be looking at a slightly different side of the Moon, and features such as craters and lava flows would be at different angles to us. This phenomenon happens when there are large-scale changes to the density distribution of a planet and is referred to as ‘true polar wander’. Independent studies have found that the Moon’s polar ice caps have shifted slightly over time due to this wander, and our prediction for the location of the magnetic north pole is consistent with that shift.
There are still many unanswered questions about the Moon’s geologic history, but our study has shown the power of working on oriented samples from other planets. This single observation at just one location on the lunar surface has significantly improved our understanding of how the ancient lunar magnetic field was generated.
We are about to enter a new dawn of planetary science with oriented samples currently being drilled on the Martian surface and due for return in the next decade, and the Artemis program aiming to send humans back to the Moon by 2024. We have shown the significance of collecting oriented samples collected in geologic context that have not moved since their formation. Our results can guide future sampling strategies so we can understand more about how the interior of the Moon has evolved over time.
The full paper, The palaeoinclination of the ancient lunar magnetic field from an Apollo 17 basalt, was published in Nature Astronomy.
 Jack Schmitt on the Moon during the Apollo 17 mission (credit NASA)
Jack Schmitt on the Moon during the Apollo 17 mission (credit NASA)The Moon’s polar regions are home to craters and other depressions that never receive sunlight. Permanently shadowed lunar craters contain water ice but are difficult to image. An AI algorithm now provides sharper images, allowing us to see into them with high resolution for the first time.
Near the lunar north and south poles, the incident sunlight enters the craters and depressions at a very shallow angle and never reaches some of their floors.
Dr. Valentin Bickel, MPS
Today, a group of researchers led by the Max Planck Institute for Solar System Research (MPS) in Germany, supported by the University of Oxford and the NASA Ames Research Center, have taken a closer look at some of these regions and presented the highest-resolution images to date covering 17 such craters in the journal Nature Communications.
Craters of this type could contain frozen water, making them attractive targets for future lunar missions, and the researchers focused further on relatively small and accessible craters surrounded by gentle slopes. Three of the craters have turned out to lie within the just-announced mission area of NASA's Volatiles Investigating Polar Exploration Rover (VIPER), which is scheduled to touch down on the Moon in 2023.
Imaging the interior of permanently shadowed craters is difficult, and efforts so far have relied on long exposure times resulting in smearing and lower resolution. By taking advantage of reflected sunlight from nearby hills and a novel image processing method, the researchers have now produced images at 1-2 meters per pixel, which is at or very close to the best capability of the cameras.
The Moon is a cold, dry desert. Unlike the Earth, it is not surrounded by a protective atmosphere and water which existed during the Moon’s formation has long since evaporated under the influence of solar radiation and escaped into space. Nevertheless, craters and depressions in the polar regions give some reason to hope for limited water resources.
'Near the lunar north and south poles, the incident sunlight enters the craters and depressions at a very shallow angle and never reaches some of their floors', MPS-scientist Dr. Valentin Bickel, first author of the new paper, explains. In this "eternal night," temperatures in some places are so cold that frozen water is expected to have lasted for millions of years. Impacts from comets or asteroids could have delivered it, or it could have been outgassed by volcanic eruptions, or formed by the interaction of the surface with the solar wind.
 An as-yet unnamed crater (region 1 in figure below) near the Moon’s south pole and close to the proposed landing site of NASA's Volatiles Investigating Polar Exploration Rover. Left shows an image taken by the Lunar Reconnaissance Orbiter. Right shows the same image after our image processing. credits: left: NASA/LROC/GSFC/ASU; right: MPS/University of Oxford/NASA Ames Research Center/FDL/SETI Institute
An as-yet unnamed crater (region 1 in figure below) near the Moon’s south pole and close to the proposed landing site of NASA's Volatiles Investigating Polar Exploration Rover. Left shows an image taken by the Lunar Reconnaissance Orbiter. Right shows the same image after our image processing. credits: left: NASA/LROC/GSFC/ASU; right: MPS/University of Oxford/NASA Ames Research Center/FDL/SETI InstituteMeasurements of neutron flux and infrared radiation obtained by space probes in recent years indicate the presence of water in these regions. Eventually, NASA’s Lunar Crater Observation and Sensing Satellite (LCROSS) provided direct proof: twelve years ago, the probe fired a projectile into the shadowed south pole crater Cabeus. As later analysis showed, the dust cloud emitted into space contained a considerable amount of water.
'With the help of the new HORUS images, it is now possible to understand the geology of lunar shadowed regions much better than before, Ben Moseley', Oxford University.
However, permanently shadowed regions are not only of scientific interest. If humans are to ever spend extended periods of time on the Moon, naturally occurring water will be a valuable resource - and shadowed craters and depressions will be an important destination.
NASA's uncrewed VIPER rover, for example, will explore the South Pole region in 2023 and enter such craters. To get a precise picture of their topography and geology in advance - for mission planning purposes, for example - images from space probes are indispensable. NASA’s Lunar Reconnaissance Orbiter (LRO) has been providing such images since 2009.
However, capturing images within the deep darkness of permanently shadowed regions is exceptionally difficult; after all, the only sources of light are scattered light, such as that reflecting off the Earth and the surrounding topography, and faint starlight. 'Because the spacecraft is in motion, the LRO images are completely blurred at long exposure times,' explains Ben Moseley of the University of Oxford, a co-author of the study.
At short exposure times, the spatial resolution is much better. However, due to the small amounts of light available, these images are dominated by noise, making it hard to distinguish real geological features. To address this problem, the researchers have developed a machine learning algorithm called HORUS (Hyper-effective nOise Removal U-net Software) that "cleans up" such noisy images.
It uses more than 70,000 LRO calibration images taken on the dark side of the Moon as well as information about camera temperature and the spacecraft's trajectory to distinguish which structures in the image are artifacts and which are real. This way, the researchers can achieve a resolution of about 1-2 meters per pixel, which is five to ten times higher than the resolution of all previously available images.
Using this method, the researchers have now re-evaluated images of 17 shadowed regions from the lunar south pole region which measure between 0.18 and 54 square kilometers in size.
In the resulting images, small geological structures only a few meters across can be discerned much more clearly than before. These structures include boulders or very small craters, which can be found everywhere on the lunar surface. Since the Moon has no atmosphere, very small meteorites repeatedly fall onto its surface and create such mini craters.
Because the spacecraft is in motion, the LRO images are completely blurred at long exposure times, Ben Moseley, Oxford University.
'With the help of the new HORUS images, it is now possible to understand the geology of lunar shadowed regions much better than before,' explains Moseley. For example, the number and shape of the small craters provide information about the age and composition of the surface. It also makes it easier to identify potential obstacles and hazards for rovers or astronauts. In one of the studied craters, located on the Leibnitz Plateau, the researchers discovered a strikingly bright mini-crater.
‘Its comparatively bright color may indicate that this crater is relatively young,’ says Bickel. Because such a fresh scar provides fairly unhindered insight into deeper layers, this site could be an interesting target for future missions, the researchers suggest.
The new images do not provide evidence of frozen water on the surface, such as bright patches. “Some of the regions we've targeted might be slightly too warm," Bickel speculates. It is likely that lunar water does not exist as a clearly visible deposit on the surface at all - instead, it could be intermixed with the regolith and dust, or may be hidden underground.
To address this and other questions, the researchers' next step is to use HORUS to study as many shadowed regions as possible. ‘In the current publication, we wanted to show what our algorithm can do. Now we want to apply it as comprehensively as possible,’ says Bickel.
This work has been enabled by the Frontier Development Lab (FDL.ai). FDL is a co-operative agreement between NASA, the SETI Institute (seti.org) and Trillium Technologies Inc, in partnership with the Luxembourg Space Agency and Google Cloud.
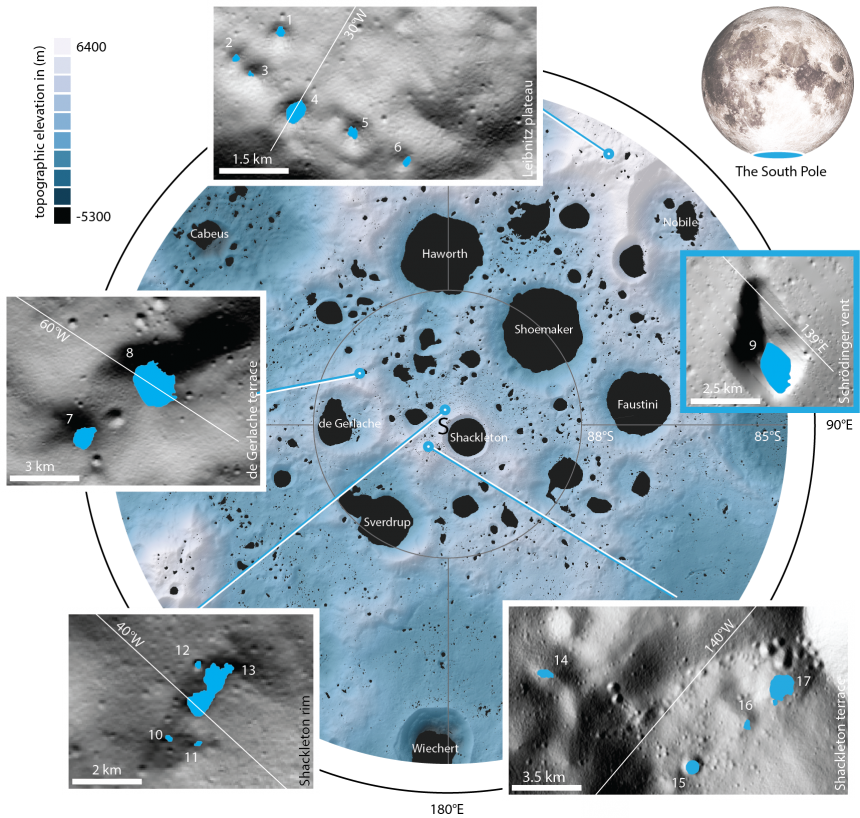 The 17 newly studied craters and depressions are located near the South Pole. Their sizes range from 0.18 to 54 square kilometres. Region 9 is not located in the section of the south polar region shown here, but a bit further to the North, in the Schrödinger Basin. credits: MPS/University of Oxford/NASA Ames Research Center/FDL/SETI Institute
The 17 newly studied craters and depressions are located near the South Pole. Their sizes range from 0.18 to 54 square kilometres. Region 9 is not located in the section of the south polar region shown here, but a bit further to the North, in the Schrödinger Basin. credits: MPS/University of Oxford/NASA Ames Research Center/FDL/SETI InstituteIn March 2020, the World Health Organization declared a global COVID-19 pandemic and the UK announced a strict national lockdown. When Oxford University scientists were sent home, they rolled up their sleeves, switched on their computers and started to help develop new drugs to target SARS-CoV-2, the virus that causes COVID-19.
‘What is remarkable about this collaboration, involving 29 scientists from around world, is that every meeting was entirely virtual, with many collaborators yet to meet face-to-face.’ Prof. Garrett M. Morris, Oxford University.
It began with a small group of scientists and their graduate students at the University of Oxford, who were joined by colleagues from Bristol University, Diamond Light Source and academics in France, Italy, and Spain – all working from home but coming together weekly on Zoom to tackle this terrifying disease.
Thus began an international collaboration involving 29 scientists around the world focused on understanding how SARS-COV-2 makes its worker proteins at the molecular level so we could develop novel antiviral drugs and block their production.
‘This collaboration has really shown how sharing of models, data and expertise can help get answers and understanding much more quickly. It’s how science should be done – particularly in the face of pressing problems like the Covid pandemic.’ Prof. Adrian Mulholland, Bristol University
Despite the development of successful vaccines in record time, there are no drugs that have been designed specifically to target COVID-19, but they are desperately needed.
Once SARS-CoV-2 has invaded a healthy human cell, the virus's own genetic material commandeers the infected cell's machinery, forcing it to make new copies of the virus.
The new virus begins as one long protein that cuts itself into functional units. First the cutting catalyst or 'protease' cuts itself out, which then cuts at multiple other positions.
‘The opportunity to apply our basic science on enzymes to a problem of enormous societal value in a fantastically collaborative manner was a wonderful life experience.’ Prof. Chris Schofield, Oxford University.
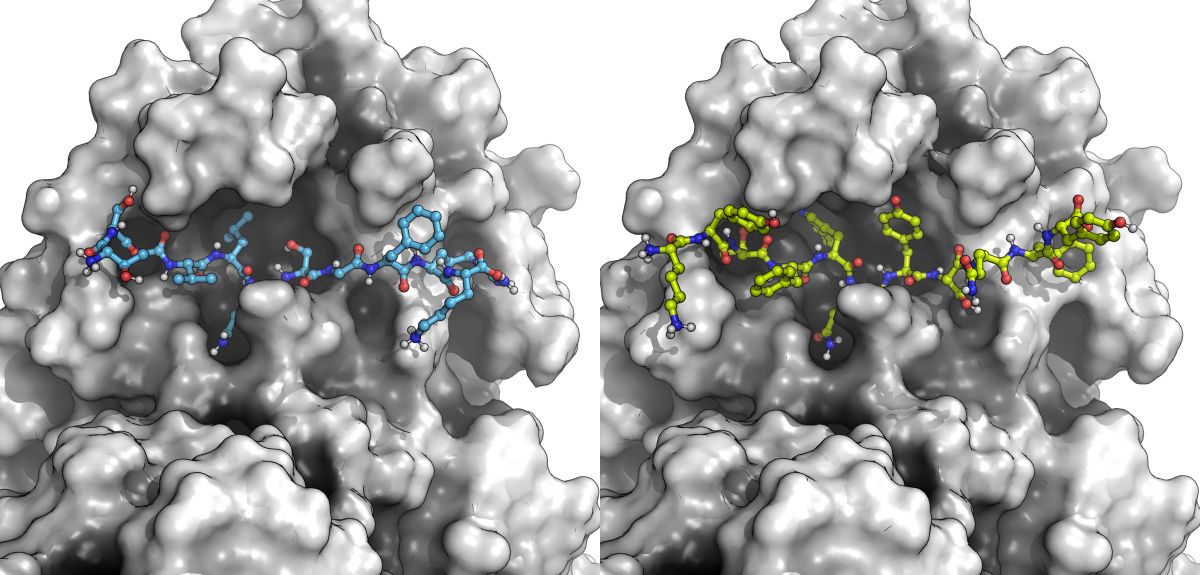
SARS-CoV-2 has two molecular machines or proteases that resemble 'molecular scissors'. One of these, called the main protease, or 'Mpro' for short, cuts at no less than 11 of these cut sites.
If scientists can design new molecules that bind more tightly than these natural substrates, they could stop the virus dead in its tracks. Blocking the cutter stops the virus from replicating—a strategy that has worked for treating other viral diseases like HIV and hepatitis.
Using 3D structures obtained by shining X-rays onto crystals of the main protease of SARS-CoV and SARS-CoV-2, Prof. Morris and his collaborators were able to develop computational models of how the SARS-CoV-2 Mpro binds to its 11 cut sites. From these models, they were able to gain key insights into how these viral Mpro 'molecular scissors' work.
Building on this knowledge, and using computational methods, they next sought to design novel molecules that could bind even more tightly than the natural cut sites. Using computers to sift through just over 200 trillion possibilities, they proposed new molecules that would stop the virus from maturing.
‘This experience taught me that science can be carried out regardless of working conditions, when a group of enthusiastic and motivated people gather together’. Luigi Genovese, CEA France.
All 11 cut sites and 4 of these designed peptides were synthesized and tested in the laboratory of Prof. Chris Schofield in the Chemistry Research Laboratory at the University of Oxford. The experiments showed that the novel designed peptides not only bound to the molecular scissors but blocked the substrates and actually inhibited the Mpro.
Scientists also carried out an extensive analysis of hundreds of published 3D structures of small molecules bound to Mpro and predicted how inhibitors designed by the COVID Moonshot would bind, figuring out how these 'molecular keys' fitted into the 'molecular lock' of Mpro, and using these insights to propose how to design new drugs to treat COVID-19.
‘Ironically, the busiest time of my PhD was when I was sitting at home during lockdown, working on the collaboration. The weekly, big zoom meetings created a great structure in an otherwise monotonous ‘lockdown-life’ schedule.’ Marc Moesser, Department of Statistics, Doctoral Student.
The team combined their expertise, applying a wide range of computational modelling techniques to build a complete picture of SARS-CoV-2 Mpro.
Much of the work was carried out by four graduate students: Henry Chan, Marc Moesser and Tika Malla at Oxford University, and Becca Walters at Bristol University.
These techniques ranged from comparative molecular modelling, molecular dynamics, interactive molecular dynamics in virtual reality, quantum mechanics, computational peptide design, protein-ligand docking, protein-peptide docking, to protein-ligand interaction analysis.
The team included: Prof. Garrett M. Morris (Department of Statistics), Prof. Fernanda Duarte, and Prof. Chris Schofield (Department of Chemistry) from the University of Oxford; Prof. Adrian Mulholland, Dr. Debbie Shoemark, Dr. Richard Sessions, and Prof. James Spencer from the University of Bristol, UK; Prof. Martin Walsh from Diamond Light Source, U.K. and Dr. Luigi Genovese of the CEA (Atomic Energy Commission) in France; and Prof. Alessio Lodola from the University of Parma, Italy, and Prof. Vicent Moliner and Dr. Katarzyna Świderek from the University of Jaume I, Spain joining the team as the project progressed.
The report, 'Discovery of SARS-CoV-2 Mpro Peptide Inhibitors from Modelling Substrate and Ligand Binding', has just been published in the Royal Society of Chemistry's flagship journal, Chemical Science.
 Some of the Scientists investigating SARS-COV-2 who only met virtually
Some of the Scientists investigating SARS-COV-2 who only met virtuallyAs the planet warms, and our population grows, there is an ever-increasing pressure on natural resources. It has been estimated that ‘stressed’ crops - from changing weather patterns, drought, flooding and extreme temperature - may reduce production by as much as 70%, which will have devastating impact on our ability to feed the world.
‘It’s becoming increasingly clear that such post-translational regulation of chloroplast proteins is vital for plant growth and productivity, and thus of course for food production’. Professor Paul Jarvis
It has become increasingly urgent that we develop improved crop varieties - plants with enhanced nutritional value or resilience to adverse environments - and key to this development will be our understanding of the molecular basis of plant stress tolerance.
New research from the University of Oxford, published recently in the journal eLife, sheds fresh light on plant chloroplasts, and the proteins inside them. The regulation of chloroplast proteins is important for plant development and stress acclimation and is increasingly significant as plants - including our staple crops, wheat, rice, barley - are having to respond to our changing environments.
‘As the planet warms, it will be increasingly urgent to understand the molecular basis of plant stress tolerance.’ Dr Samuel Watson
All green plants grow by converting light energy into chemical energy via a process known as photosynthesis. Photosynthesis occurs within specialised compartments of plant cells known as chloroplasts. Chloroplasts require thousands of different proteins to function, and these are imported into the chloroplast via a specialised machinery known as the TOC complex. The TOC complex is, itself, made of proteins.
Recent studies revealed that the TOC complex is rapidly destroyed when plants encounter environmental stress - this protects plant cells from damage by limiting photosynthesis, which can generate toxic by-products under adverse conditions. This process has been named CHLORAD, for “chloroplast-associated protein degradation”.
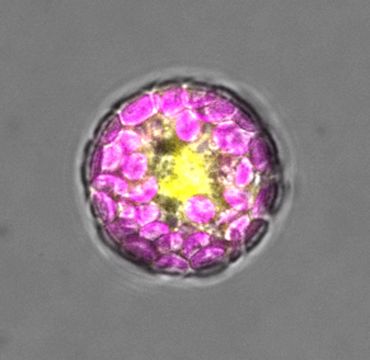 Plant cell (a protoplast) expressing the SUMO1 protein tagged with yellow fluorescent protein (YFP); the chloroplasts inside the cell are coloured magenta in this image
Plant cell (a protoplast) expressing the SUMO1 protein tagged with yellow fluorescent protein (YFP); the chloroplasts inside the cell are coloured magenta in this imageIn CHLORAD, the TOC complex is first marked with a small protein called ubiquitin. This ‘ubiquitination’ promotes the destruction of the complex, and thus suppresses chloroplast protein import, photosynthesis, and the production of toxic by-products.
‘This study has uncovered another layer of complexity within the systems that plants use to control their chloroplasts.’ Professor Paul Jarvis
In this study, researchers asked whether the TOC complex is also SUMOylated - SUMO is another small tag that is similar to ubiquitin - and, if so, what the function such TOC SUMOylation is.
The researchers found that the TOC complex is indeed SUMOylated, and that TOC SUMOylation also triggers the destruction of the TOC complex and is important for plant growth and development. These results are intriguing, as they indicate that SUMO action is very similar to that of CHLORAD, in this context.
In fact, the observed similarity with CHLORAD implies that SUMOylation regulates the activity of the CHLORAD pathway. This is particularly interesting, as SUMOylation is known to be induced by various forms of environmental stress and is a key driver of plant stress acclimation.
Professor Paul Jarvis, who supervised the work said: ‘It was remarkable when the role for ubiquitination, and CHLORAD, was discovered a few years ago, and this new role for SUMO just adds to the intrigue.’
Building on these discoveries, the researchers are currently exploring how the CHLORAD pathway can be manipulated to improve crop performance. Better understanding of the regulation of chloroplast protein import and/or the CHLORAD pathway, delivered as a result of the new findings reported here, will help to guide these efforts.
 Prof. Jonathan Doye
Prof. Jonathan DoyeMost people who have done some chemistry will have seen examples of how the directionality of the covalent bonding for a given element can determine the crystal structures they form. For example, the tetrahedral bonding of carbon atoms naturally leads to the crystalline structure of diamond. But to better understand this challenge, we first need to explain what a quasicrystal is.
We should start with a discovery of Sir Roger Penrose, an Oxford mathematician and physicist who won the Nobel Prize for Physics in 2020. In 1974 he showed that one could tile a plane with just two types of tiles, such that the tiling has no repeating pattern but has a kind of 5-fold symmetry that is not possible for crystals. These Penrose tilings, one of which is shown below, are said to have “quasiperiodic order”.
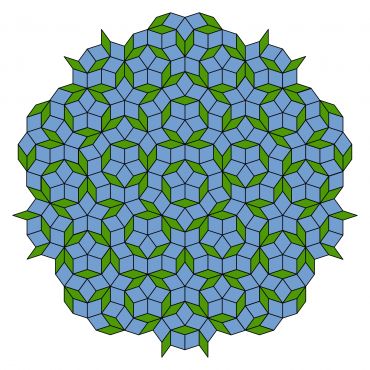 An example of a Penrose tiling
An example of a Penrose tiling
The order of these quasicrystals is revealed by the diffraction patterns that result when electrons or x-rays pass through them. They are labelled as icosahedral, because like the icosahedron that is one of the Platonic solids, there are six axes along which five-fold symmetry is revealed in the diffraction patterns. Like crystals, this underlying symmetry can also be revealed by the shapes into which quasicrystals grow, such as the dodecahedron below. Dan Shechtman’s paradigm-shifting discovery led to him receiving the Nobel Prize in Chemistry in 2011.
 A large quasicrystal with a docahedral shape that is a consequence of its underlying non-crystallographic icosahedral symmetry
A large quasicrystal with a docahedral shape that is a consequence of its underlying non-crystallographic icosahedral symmetryThis discovery led to an intense search for other examples of icosahedral quasicrystals. Many were found, including even natural examples in meteorites, but all were metallic alloys, whereas none have been found in covalent materials where the bonding is directional.
This raises the question of whether, if one is not restricted by the geometry of covalent bonding that is allowed by atoms, it might be possible to design particles that could form icosahedral quasicrystals through directional bonding. In 2014 Eva González Noya, my collaborator at CSIC in Madrid, and myself set ourselves the task of finding computational models that could achieve this.
One of the challenges we faced is that the structure of quasicrystals is more complex to describe than periodic crystals. For example, if one has the diffraction pattern for a crystal, one can work out exactly where all the atoms are in the unit cell. However, for an icosahedral quasicrystal, one instead gets a description of the quasicrystal in terms of a crystal in six dimensions!
Some help came from computational experiments by the group of Sharon Glotzer at the University of Michigan in which icosahedral quasicrystals formed in systems without directional bonding. Although these provided examples where the positions of all the particles were known, they revealed another challenge: although the icosahedral quasicrystals have beautiful long-range order, locally they are quite disordered. For example, if one was to try to have particles that reproduce all the different patterns of local bonding in this example, the number of types of particles needed would be far too large to be practical.
Progress was gradually made, a key insight being that one should not be too fussy about whether a particle is able to form all its possible directional bonds, and in 2018 a first example system consisting of five particle types was found that assembled into an icosahedral quasicrystal in simulations; later, this was simplified to a system of two particle types.
And to show the generality of the design principles, a second quasicrystal-forming system was developed that had a totally different pattern of local directional bonding. Somewhat like the metallic alloy examples, key building blocks are icosahedral clusters (the yellow and cyan rhombic triacontahedra in the image below) which form in a matrix of particles that propagates the global order.
 A close-up of a cut through one of the icoshedral quasicrystals grown in our computational experiments viewed along a five-fold axis of symmetry.
A close-up of a cut through one of the icoshedral quasicrystals grown in our computational experiments viewed along a five-fold axis of symmetry.Of course, an important question is whether these computationally discovered quasicrystals could be realized experimentally. Although it is unlikely that they could be formed by atomic materials, atoms are not the only particles that form crystals. Nanoparticles and colloidal particles can also form crystals; for example, precious opals are crystals of colloidal silica spheres and their iridescence results from the diffraction of light by these crystals.
Recently, crystals made up from building blocks made of DNA have been realized. The beauty of these DNA nanotechnology techniques is that the shape of the DNA particles and the interactions between them can be almost arbitrarily controlled. Computational modelling of such DNA particles suggests that making icosahedral quasicrystals from DNA origami particles could well be feasible.
How to design an icosahedral quasicrystal through directional bonding, Published 18th August 2021, Nature.
- ‹ previous
- 8 of 9
- next ›




 25-year Oxford study finds the effects of conflict last for generations
25-year Oxford study finds the effects of conflict last for generations What Louise Thompson’s campaign tells us about the national maternity crisis
What Louise Thompson’s campaign tells us about the national maternity crisis Celebrating 25 Years of Clarendon
Celebrating 25 Years of Clarendon  Learning for peace: global governance education at Oxford
Learning for peace: global governance education at Oxford  What US intervention could mean for displaced Venezuelans
What US intervention could mean for displaced Venezuelans  10 years on: The Oxford learning centre making an impact
10 years on: The Oxford learning centre making an impact Oxford and The Brilliant Club: inspiring the next generation of scholars
Oxford and The Brilliant Club: inspiring the next generation of scholars New course launched for the next generation of creative translators
New course launched for the next generation of creative translators