Image: Shutterstock
The puzzle of plasmids
We all know that antibiotic-resistant bacteria are a problem – maybe even 'as big a risk as terrorism'* – but how is this resistance passed on from one bug to another?
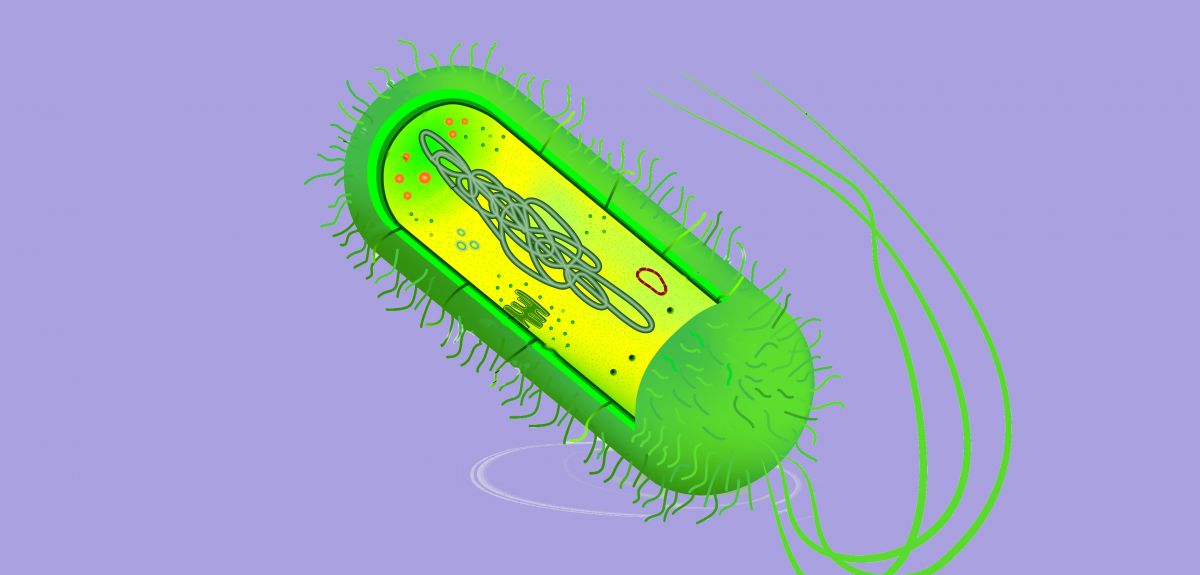
One key culprit is plasmids: these are small 'hula hoops' of DNA that occur naturally within the cells of bacteria (and some eukaryotes) and are completely independent of the main bacterial genome.
'Plasmids carry genes that help bacteria to adapt to new niches and stresses, playing a key role in bacterial evolution,' said Alvaro San Millan of Oxford University's Department of Zoology. 'In the last decades they have become especially relevant as vehicles for the spread of antibiotic resistance. Understanding the biology and evolution of plasmids is key to controlling the global threat posed to human health by antibiotic resistance.'
Not only do plasmids replicate, copying across to a daughter cell when a bacterium divides, but they can also pass between unrelated bacteria through mechanisms such as cell-to-cell contact or a 'bridging connection' in a process called 'conjugation' – which has been described as the bacterial equivalent of sex.
Alvaro explains that plasmids usually transfer not just one but a number of ways to fight off antibiotics: 'they confer multi-resistance en bloc, in only one step, jeopardising the effectiveness of antibiotic therapy.'
But there are a couple of reasons why plasmids shouldn't be such a problem:
For a bacterium, carrying a plasmid around is a bit like having a free-loading lodger. In situations where the lodger is providing a useful service, such as protecting you from antibiotics when antibiotics are killing off your rivals, then it can give you an advantage. Take those antibiotics away, however, and the evolutionary costs begin to mount so that a plasmid-free bacterium should fare better.
And then there's conjugation: a recent analysis has shown that almost half of the plasmids in nature lack the genetic tools required for conjugation. So how do non-conjugative plasmids survive?
Alvaro and colleagues set out to investigate this plasmid puzzle using a combination of mathematical modelling and experimental evolution and report their findings in a recent article in Nature Communications.
What they found is that, after exposure to antibiotics, bacteria evolve compensatory adaptions to mitigate some of the cost of carrying a plasmid. The adapted plasmid-carriers are selected in the next exposure to antibiotics and when the antibiotic is removed, plasmids are eliminated much more slowly from the population because they no longer come with a cost. Any subsequent exposure to antibiotics will cause the population of plasmid-carriers to bounce back as their resistance once again becomes an advantage, increasing their chances for further adaptations.
'It appears that adaptation between plasmid and bacteria coupled with rare events of antibiotic treatment are enough to stabilise a non-conjugative plasmid in a bacterial population,' Alvaro tells me. 'Our results provide a new understanding of how plasmids can persist in bacterial populations and help to explain why plasmid-mediated resistance can be maintained after antibiotic use is stopped.'
He adds: 'Our paper shows how antibiotic use plays a key role in stabilising resistance genes in pathogen populations, highlighting the importance of minimising antibiotic use for controlling resistance.'
The researchers say that more studies are needed into how plasmids evolve and survive in bacterial populations so that we can design intervention strategies aimed at reducing the spread of antibiotic resistance.
*In the opinion of the UK’s chief medical officer, see this BBC report.
A report of the research, entitled 'Positive selection and compensatory adaptation interact to stabilize non-transmissible plasmids', is published in Nature Communications.
 Genetic insights into our evolutionary battle with malaria
Genetic insights into our evolutionary battle with malaria Promising results in efforts to develop new malaria treatments
Promising results in efforts to develop new malaria treatments Unfolding role of cell's gatekeepers
Unfolding role of cell's gatekeepers Online help to democratise global health research
Online help to democratise global health research Shootout at the OK Colon
Shootout at the OK Colon