Vibrations reveal how material 'takes a breath'
A combination of supercomputer calculations and a bombardment of high-energy particles has revealed how a new kind of material opens its pores and 'breathes'.
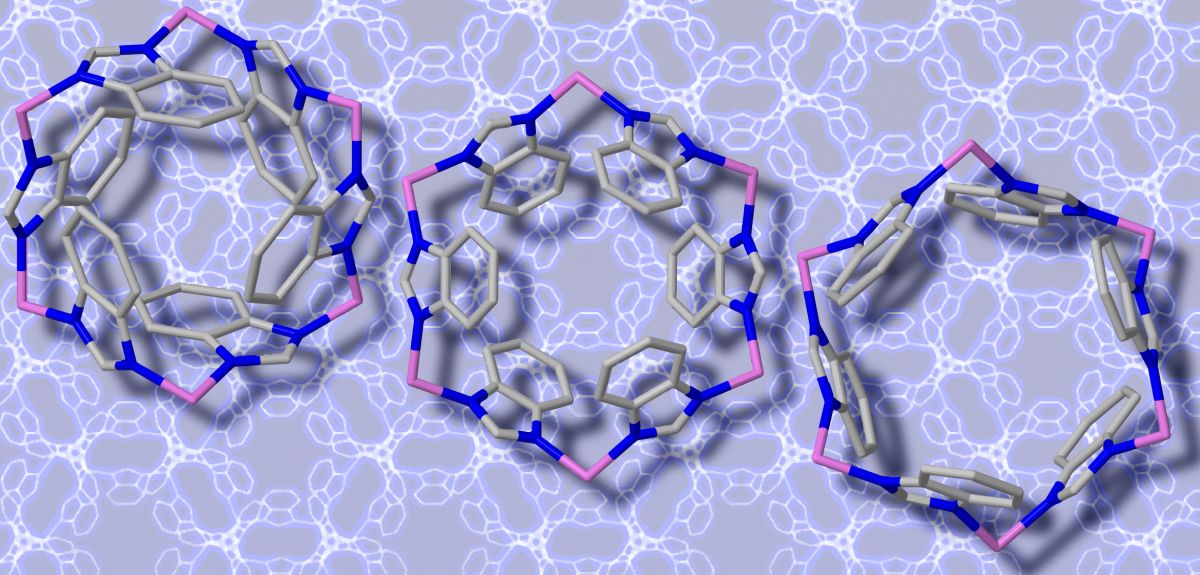
Metal-organic frameworks (MOFs), are formed from building blocks made up of metal ions connected by organic molecules. These molecular blocks assemble themselves to produce a variety of crystal-like structures whose porous nature and ‘shape-shifting’ abilities could make them ideal for emerging applications, such as trapping greenhouse gases or delivering drugs.
'One of the key selling points of MOFs is the exceptionally large internal surface area that some of the frameworks can possess,' Matthew Ryder, a DPhil student in the Multifunctional Materials & Composites (MMC) Laboratory at Oxford University's Department of Engineering Science, tells me. 'Some MOFs have internal surface areas as large as 10,000 square metres per gram and to put that into perspective, that's a larger surface than a football field in every gram of MOF material!'
Whilst MOFs are similar to traditional microporous materials, such as inorganic zeolites or the activated carbons used to filter drinking water or air, they typically have a surface area ten times greater and can be processed at much lower temperatures. MOFs can also be built from a wider range of metal ions and organic links so that the desirable characteristics, such as pore size and its functionality, can be 'fine-tuned'.
Unlike activated carbons, MOFs are highly crystalline and this means that their 3D crystal structure can be precisely determined using diffraction techniques, such as X-rays and neutrons. Accurate 3D representations of MOF structures are central to computational modelling studies.
'It has been suggested for some time that the practical functionalities of each specific MOF material is intrinsically controlled by its elastic responses and collective vibrations of the porous framework (called 'lattice dynamics') down at the molecular scale,' Matthew explains.
In a study recently published in Physical Review Letters the team, led by Oxford's Professor Jin-Chong Tan, reports a new method for investigating how MOFs vibrate. They tested their ideas on a subclass of MOF materials: Zeolitic Imidazolate Frameworks (ZIFs).
Their method used Density Functional Theory (DFT) to unravel the complete vibrational nature of the frameworks at the molecular level. These calculations were so demanding that they could only be accomplished on state-of-the-art supercomputers (at ARC in Oxford, SCRAF at Rutherford Appleton Laboratory in Harwell, and the SuperMUC Petascale System near Munich). The theory was then confirmed using high-resolution spectroscopic experiments at Diamond Light Source and the ISIS Pulsed Neutron & Muon Source at Harwell, Oxford.
The team found that the experiments closely matched the theoretical DFT predictions across the entire vibrational spectra and discovered that the most exciting MOF framework vibrational behaviour was located in the low-energy or 'Terahertz (THz) region'.
'We demonstrated for the first time that the Terahertz modes not only show the standard lattice vibrations, but also reveal all of the physical characteristics unique to the specific MOFs we studied (ZIF-4, ZIF-7 and ZIF-8),' Matthew tells me.
'Our results revealed intriguing Terahertz vibrational modes [watch animations here], which include co-operative 'gate-opening' and 'breathing' of the nano-sized pores of MOFs, crucial for the understanding of gas separation, storage, and sensing.
'Significantly, this study enabled us to gain new insights into mechanical properties of MOFs, elucidating possible phase change mechanisms (called 'soft modes') through which the porous framework may destabilise, distort or even collapse when subject to mechanical forces, thereby completely destroying their functionality. Furthermore, soft modes may give rise to anomalous and counter-intuitive mechanical behaviour, such as negative thermal expansion and auxeticity.'
By studying the Terahertz vibrations in MOFs the researchers believe they could pinpoint and overcome deformation mechanisms that could otherwise make them difficult to use commercially.
Understanding how MOFs vibrate, change shape, and 'breathe', could also make it possible to enhance how they trap specific gas molecules – such as greenhouse gases – and help to tailor them for the targeted delivery of anti-cancer drugs.
'Interestingly, the latest research into MOFs has concentrated on other less conventional applications of porous materials: everything from microelectronics and information storage, to water splitting for sustainable hydrogen production and solar energy harvesting (photovoltaics) for clean electricity generation,' Professor Tan comments.
'Engineers, materials scientists and chemists have a big role to play to ensure the future success of MOFs. Discovering more about the mechanical properties and long-term durability of these materials will be key to realising their full potential and making the leap from the laboratory into large-scale commercial applications.'
 Halloween special: zombies vs maths
Halloween special: zombies vs maths Brain stimulation for your stammer
Brain stimulation for your stammer The puzzle of plasmids
The puzzle of plasmids Learning to live with bears
Learning to live with bears Genetic insights into our evolutionary battle with malaria
Genetic insights into our evolutionary battle with malaria Learning machines turn to quantum
Learning machines turn to quantum